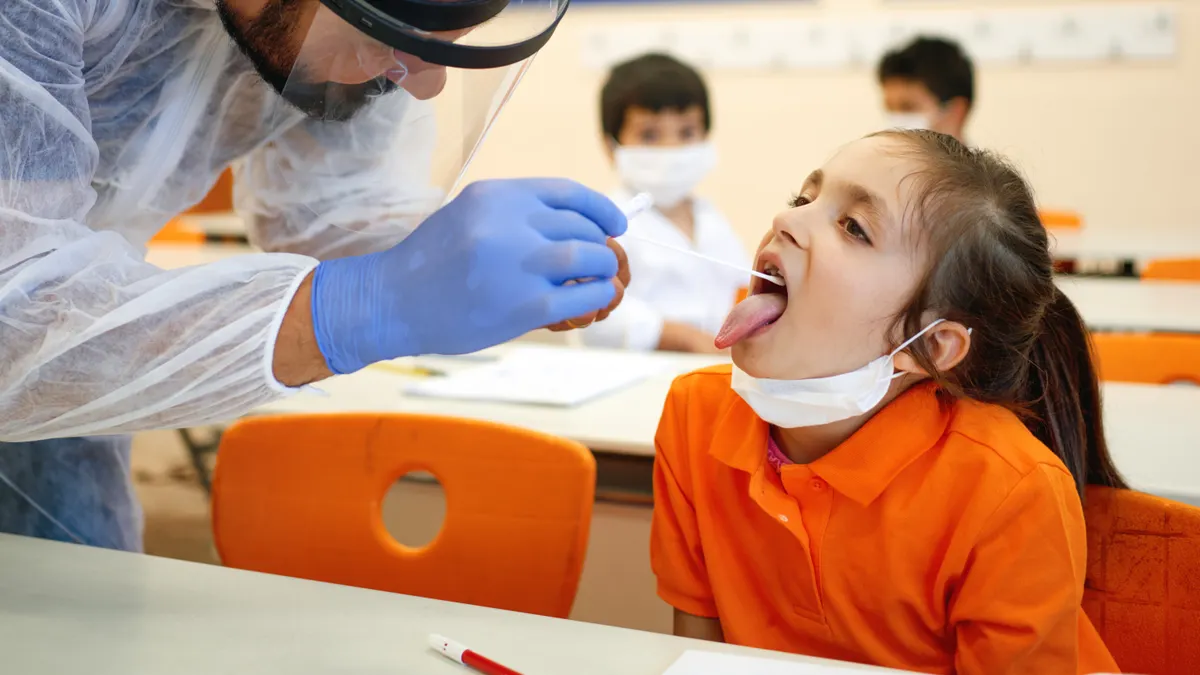
Educators, parents, and students all know how important tests are to the school experience. And testing for COVID-19 is no different.
As part of the push under President Joe Biden to reopen schools, the administration recently announced that it would make $10 billion available for K-12 schools to expand coronavirus testing of students, teachers, and staff.
Public health experts are optimistic widespread vaccination will drive cases lower but testing still remains critical in detecting new cases of COVID-19.
Amid the many questions K-12 educators have around COVID-19 testing and the safety of its institutions, Dr. Charles Cooper of BD, a leading global medical technology company, shared his knowledge on the topic.
When will it be safe for students, teachers and staff to return to school?
It is finally possible to get kids back to school, but only if the proper precautions are in place.
There is a false sense of security that is coming with the decline in COVID-19 case numbers and the increasing number of vaccinations, but we are still very much battling the coronavirus pandemic. Students, teachers and staff should still wear masks and wash their hands, but school leaders and administrators have an opportunity to use rapid antigen testing as another tool to help protect their communities.
How can testing help stop the spread of COVID-19?
Data shows that there is a low risk of spreading the virus from person to person if preventative measures are put in place. Articles and studies in multiple peer-reviewed publications including the New England Journal of Medicine and the British Medical Journal have touted the benefits of serial, rapid antigen testing. In addition, a recent landmark RADx-funded study demonstrated that the serial use of diagnostic tests (used at least twice per week), including rapid antigen tests, increased the ability to detect infection.
On-the-spot rapid antigen testing can help stop the spread of COVID-19 in schools before it starts. The BD Veritor™ Plus System can issue a positive or negative test result in as little as 15 minutes, so if a student or teacher tests positive, they can be quickly isolated from the rest of the student body and the virus becomes easy to monitor.
What is the difference between rapid antigen testing and PCR testing?
PCR testing is highly sensitive, which is a strength. However, it is also more expensive and requires a laboratory setting with skilled laboratory technicians. The timeframe to get results back is also dependent on the ability to get a collection sample to a lab, and that lab has to work through its large amount of volume.
In contrast, antigen testing, is very simple to use. It's a diagnostic test that can be used by people who are not highly skilled laboratory technicians. It also has a turnaround time that's much faster. Antigen testing provides a 15-minute turnaround, so you get a result while the individual being tested is still present. It's also less expensive. Rapid antigen testing delivers a combination of simplicity, speed and reliable results that make it an ideal choice for use in K-12 schools.
How should rapid antigen testing be used in schools?
Tests like the BD Veritor™ Plus System can be used for the screening for COVID-19 through serial testing. Asymptomatic individuals like students, teachers and staff should be tested twice over two or three days with at least 24 hours and no more than 48 hours between tests.
How do we know rapid antigen testing in schools will help stop the spread?
Across the country, K-12 schools and universities have partnered with BD to implement testing programs that instill a sense of confidence that it is safe to return to school and resume in-person learning. As one example, the state of Delaware is leveraging the speed and accuracy of BD Veritor™ Plus System for rapid antigen testing in more than 76 district, charter and private K-12 institutions statewide. This scalable, in-person, low-resource program has helped enable in-person learning in the state since this past January.
What are some other things to consider when implementing a testing program at schools?
There are two ways to implement testing programs in schools. BD, for example, works directly with schools to provide step-by-step guidance on how to implement a testing program, as well as training on the BD Veritor™ Plus System, so they can manage the testing program themselves. Consolidating the lessons and strategies adopted by schools across the U.S. that have already implemented testing programs, as well as best practices from the CDC, BD works with school administrators to build out a workable, cohesive plan to implement testing.
Schools can also utilize third-party testing companies that can deliver a turnkey solution. This can alleviate the burden off school administrators and staff in managing the testing process.
Dr. Charles Cooper, M.D. is vice president for Medical and Scientific Affairs at BD, a leading global medical technology company. To learn how BD is supporting back-to-school programs, visit bdveritor.com. You can also download BD's back-to-school diagnostic testing guide, here.
- This product has not been FDA cleared or approved; but has been authorized by FDA under an EUA for use by authorized laboratories;
- This product has been authorized only for the detection of proteins from SARS- CoV-2, not for any other viruses or pathogens; and,
- This product is only authorized for the duration of the declaration that circumstances exist justifying the authorization of emergency use of in vitro diagnostics for detection and/or diagnosis of COVID-19 under Section 564(b)(1) of the Federal Food, Drug and Cosmetic Act, 21 U.S.C. § 360bbb-3(b)(1), unless the declaration is terminated or authorization is revoked sooner.
- Testing is limited to laboratories certified under the Clinical Laboratory Improvement Amendments of 1988 (CLIA), 42 U.S.C §263a, that meet the requirements to perform moderate, high or waived complexity tests. This test is authorized for use at the Point of Care (POC), i.e., in patient care settings operating under a CLIA Certificate of Waiver, Certificate of Compliance, or Certificate of Accreditation. For more information, please see bdveritor.com.